Impact Of Different Atmospheric Conditions On Metals

The speed of corrosion reaction on metal substrates is highly dependent on the environment. Various environments where coated metals are used are classified as per the reduction in the thickness or mass of the metals and metal alloys when they are subjected to different atmospheric conditions. As per the standard EN ISO 12944-2 -1998, the atmospheric conditions that corrode the metals are:
| Industrial Atmosphere | Marine Atmosphere | Country Atmosphere | |
| Pollution | High Pollution | Low Pollution | Hardly Any Pollution |
| Wet Time | Long Wet Time, High Humidity | Varies from low to high | Low Wet Time |
| Rate of Corrosion | Corrosion Rate of Zinc Plating >1.5 μm/year | Varies with Temperature, Wet Time and Relative Humidity | Corrosion rate of zinc often< 0.4 μm/year |
| Content of Salt in Atmosphere | High Content of Salt | High Content of Salt | Low Content of Salt in Atmosphere |
Types of Corrosion & Preventions
The mentioned environmental factors affect corrodes the metals with different types of corrosion. Some types of Corrosion are mentioned below:
- Edge Corrosion – Edge Corrosion is one of the examples of galvanic corrosion. It occurs mainly at the cutting edge of the galvanized steel where the cross section of the paint coated metals is subjected to the external or environmental factors. The cross-section of the area of the material is smaller than the steel substrate it means that the zinc coating on the material is not good enough to prevent corrosion. Hence, the cutting edge corrosion is quite common on the coated steel. The corrosion protection can be enhanced by adjusting the thickness of the zinc coating.
Prevention – The highly effective way to prevent the edge corrosion is a paint layer after cutting the material.

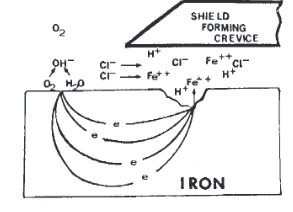
- Crevice Corrosion – This type of corrosion usually occurs on the iron material. Severe localized corrosion that often occurs within plated area of the material is known as crevice corrosion. This kind of corrosion is occurred by surface defects, deposits or dirt, holes that allow stagnant corrosion to be formed on the surface of the material. The hole of the defect on the surface is sometimes wide enough that allows the moisture or any acid to enter and corrode the material badly.
Prevention – This type of corrosion can be treated by ensuring the quality of the plating or coating so that the crevice will not form between the substrate and the coating.
- Filiform Corrosion – This occurs below the protective painted layer and can be observed as thin filament randomly distributed on the surface of the steel substrate, This type of corrosion usually weaken the material and reduces the functionality and cause an aesthetic The filament on the material initiates from the edge and move ahead to form long filaments with an active corrosion tail. This type of corrosion is usually seen on the aluminum substrates.
Prevention – This type of corrosion can be treated with the coating with very low moisture permeability. Zinc coated steel is also less prone to this kind of corrosion defect.

Apart from this, various types of corrosion are there that damage the quality of the metal substrates.
How can the Resistance of Plating to Corrosion be detected?
Although there are so much of preventions to the corrosion, still it is quite difficult for the manufacturers to ascertain that how much a metal substrate is resistant to corrosion? But this can be tested only when the material is subjected to different atmospheric conditions for a specific period of time to evaluate the actual working life of the material during their actual usage. This can be done only with the help “Salt Spray Chamber”.
The instrument is designed as per standard ASTM B117. According to this standard, the device is capable enough to create a salty environment, high temperature from ambient to 45 degree C, humidity, and moist conditions inside the chamber. The device is used to set the time of device for specific days and hours in order to ascertain the effect of different atmospheric conditions in years. This also ensures that the instrument will offer precise test results. Read more about this at Salt Spray Chamber.
Recent News
- Paper & Packaging Testing Instruments
- Paint, Plating & Coating Testing Instruments
- Plastic & Polymer Testing Instruments
- Environmental Testing Chambers
- PET & Preform Testing Instruments
- Color Measuring Testing Instruments
- View Entire Range Instruments

Catalogue 2023
Get information about new product launches, research, innovation and endeavors at Presto.
download Free CopyNeed more information
Connect with us for your business enquiries. Generally we respond within one or two working days.
send enquiriesContact Us
Quick quote
